
DISCUSS (1 of 2):
Do you think there could really be a potion that does something amazing or valuable? (Do you think there are really liquids or mixtures that can transform things?) Why or why not?
DISCUSS (2 of 2):
If you could make a potion, what would you want it to do?

GET A SUPPLY:
Everyone get 1 dull, brown penny. Then:
DISCUSS:
Suppose you wanted to make this dull, brown penny bright and shiny. Can you think of any liquids in your house that might do that?
Why do you think those liquids might work?













DISCUSS:
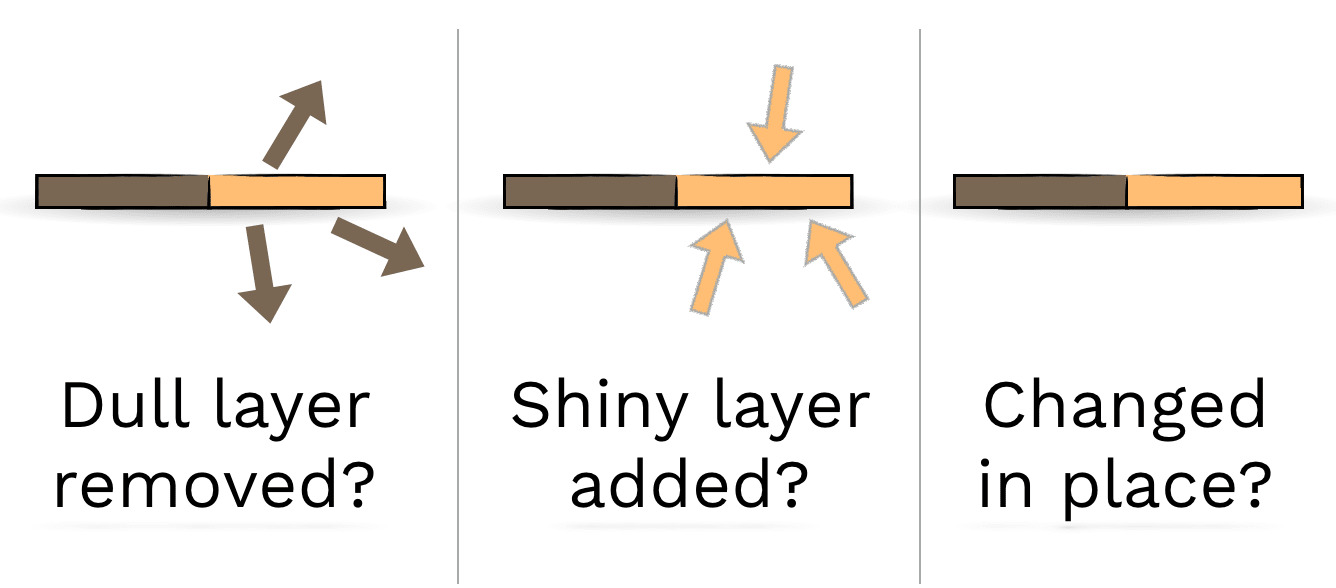
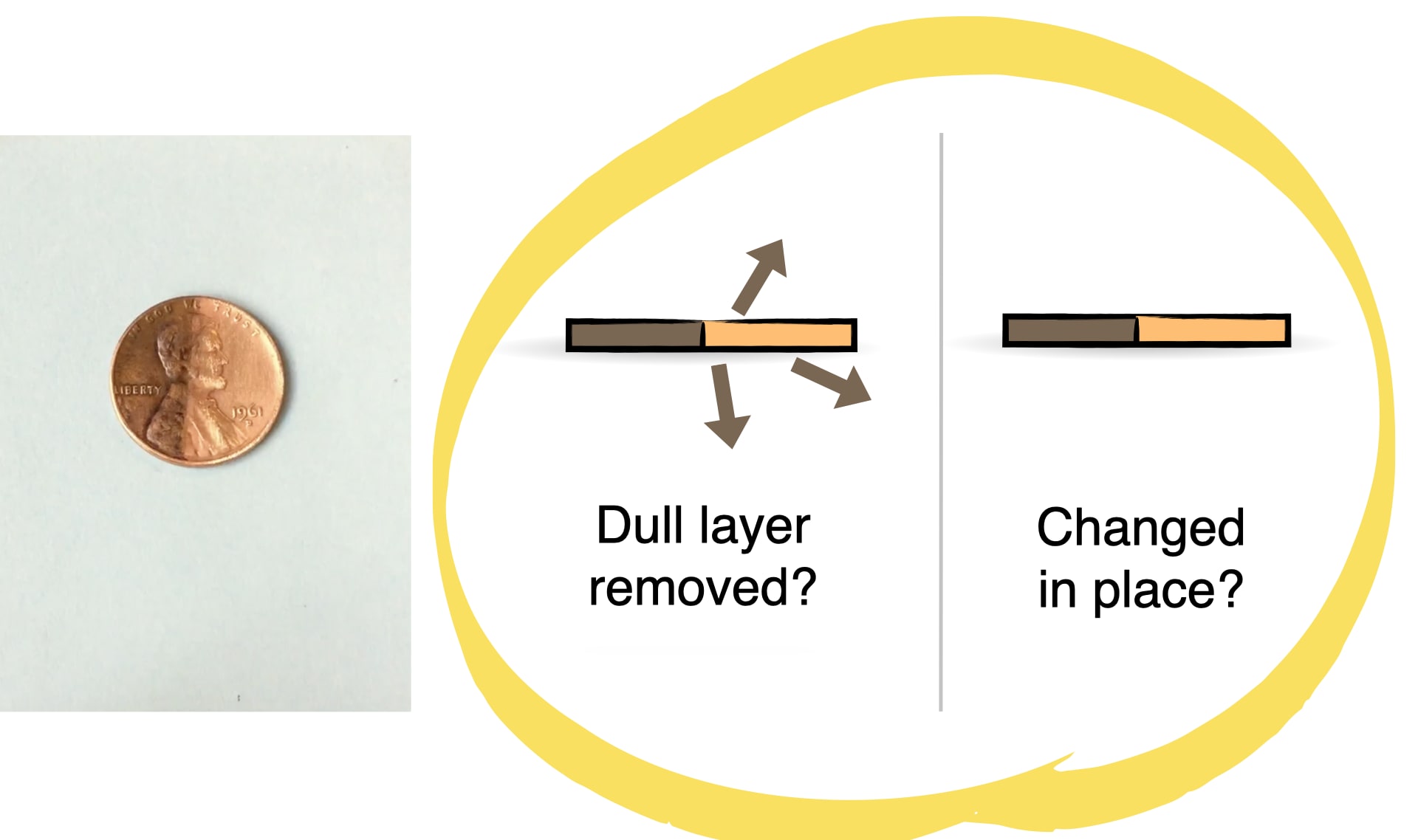
Do you think oxygen turns the penny dark brown all the way through, or just on the surface? How could you figure it out?

DISCUSS:
How could you figure out which of these three ideas is true?















Grade 5
Chemical Reactions & Properties Of Matter
Conservation of Matter
5-PS1-1, 5-PS1-2
| Alchemist’s Potion, Part 1 worksheet | 30 copies |
| Container Labels printout | Print 1 copy |
| Test Like An Alchemist printout | Print 8 copies |
|
Clean-up Supplies (Eg. Paper Towels)
|
1 roll |
|
Liquid Soap
Can also use liquid detergent.
|
Details
2 tablespoons
|
|
Scotch Tape
|
1 roll |
|
Measuring Cup
|
1 cup |
|
Measuring Spoons
|
1 set |
|
Plastic Containers w/ Lids
Each container needs to hold just over 3 cups.
|
Details
4 containers
|
|
Salt
You’ll need another cup of salt for Mystery 3, so we suggest getting a 26 oz container.
|
Details
1 cup
|
|
White Vinegar
You will also need vinegar for other lessons in this unit, so we suggest getting a gallon.
|
Details
4 cups
|
|
Pennies (must be pre-1982)
Each student needs one penny, but if you're working in a small group you need a minimum of 20. We suggest having a few extra in case some get lost.
|
Details
30 coins
|
|
Steel Nails
It’s important that you get steel nails (or steel washers)—NOT stainless-steel and NOT galvanized steel. For Mystery 2, each group of 4 students will also need one nail.
Jumbo paper clips will also work, but the results are harder to see.
|
Details
1 nail
|
We suggest students work in groups of four. Homeschool students can work on their own.
You will need access to water for this activity.
You will need old, tarnished pennies for this activity. You must use pennies dating from BEFORE 1982, when they were made from 95% copper. (Pennies made after 1982 are copper-plated zinc, which won’t work for this activity.)
We suggest asking students to bring in pennies made before 1982. You can also buy penny rolls at the bank. We bought $3 worth, sorted out the pre-1982 pennies, and had exactly 40 to work with. If you are working with a homeschool student or small group, you’ll need a minimum of 20 pennies.
Optional: Each student can use orange and brown colored pencils or crayons to show the coloration of the dull and shiny pennies on their worksheets.
In the next lesson, you’ll need to reuse some of the materials from this activity so students can copper plate a steel nail. See instructions below.
Cut out the Container Labels and tape one to each of your four plastic containers. You now need to prepare the four testing solutions - one for each container. If you’re working with a homeschool student or small group, you can cut the following “recipes” for each solution in half.
We recommend placing each container in a separate area of the classroom as a test station.
Save student work: Students will need their completed “Alchemist’s Potion, Part 1” printouts for the next lesson. Make sure they’re stored somewhere safe.
Save the pennies in the Salt & Vinegar solution: At the end of this lesson, you’ll dump all the pennies into the Salt & Vinegar to soak overnight. (If you made just 1 cup of Salt & Vinegar, dump at least 20 pennies into it.)
Add a nail: After students have left class, we recommend that you put a nail into the Salt & Vinegar solution with the pennies — but don’t tell your students you’re doing it. You’ll find out why in the next lesson, when your students will discover that the solution the pennies soaked in can change steel in a surprising way.
Grade 5
Chemical Reactions & Properties Of Matter
Conservation of Matter
5-PS1-1, 5-PS1-2
Thanks for your feedback! If you have a question or need help, please contact us. Please consider sharing your review:
Sorry the lesson didn’t go well. We read every single review in an effort to improve our Mysteries.
Thanks for letting us know. We’ll wait to ask you for feedback until after you've actually taught it.
Thanks for the feedback! We read every single review in an effort to improve our Mysteries.
Please follow these steps:
Locked
6:10

Why is the sky blue?
Locked
4:41

Why do we call them doughnuts?
Locked
5:16

Could a turtle live outside its shell?
Your membership is expired. The archive of past Mini Lessons is not included in your limited access.
View pricing